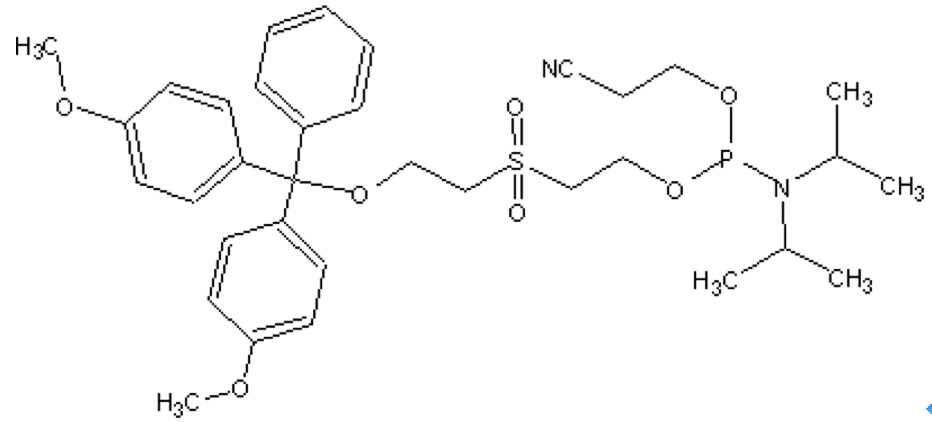
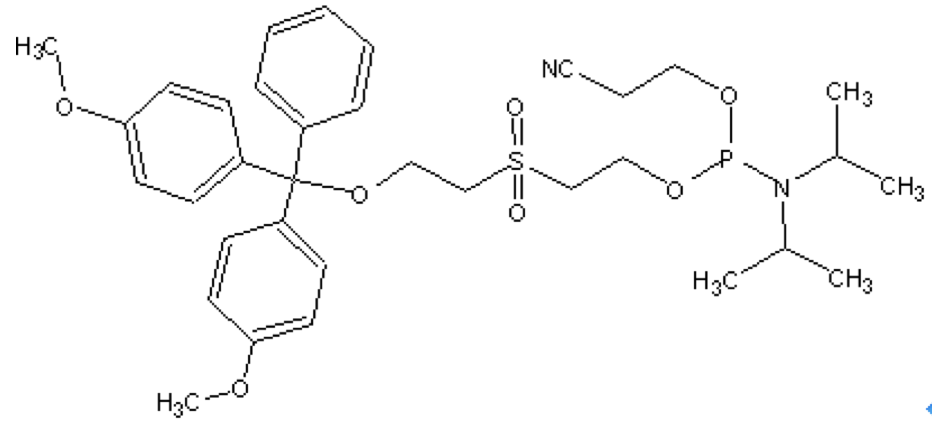
Chemical Phosphorylation Reagent I (CPR I)
Cat# | Size | Price | MW | Abs | Em | Soluble in | Storage |
6001 | 1 g | $395 | 656.77 | N/A | N/A | MeCN | F/D/L |
|
Features and Biological ApplicationsThis reagent can be used to create the 3'-phosphate by adding it as the first addition to the support in Labeling Oligonucleotides with Fluorescent Dyes. After ammonia deprotection the oligo will have the 3'-phosphate attached to the 2nd base added during synthesis. Both the support base and the CPR are cleaved. The DMT-group is removed during the ammonium hydroxide deprotection and thus is not available for poly-pak purification. |
References
1. Xu Y, Lee SA, Kutateladze TG, Sbrissa D, Shisheva A, Prestwich GD. (2006) Chemical synthesis and molecular recognition of phosphatase-resistant analogues of phosphatidylinositol-3-phosphate. J Am Chem Soc, 128, 885.
2. Ohkubo A, Ezawa Y, Seio K, Sekine M. (2004) O-selectivity and utility of phosphorylation mediated by phosphite triester intermediates in the N-unprotected phosphoramidite method. J Am Chem Soc, 126, 10884.
3. Tsuruoka H, Shohda K, Wada T, Sekine M. (2000) Synthesis and conformational properties of oligonucleotides incorporating 2'-O-phosphorylated ribonucleotides as structural motifs of pre-tRNA splicing intermediates. J Org Chem, 65, 7479.
4. Olejnik J, Krzymanska-Olejnik E, Rothschild KJ. (1996) Photocleavable biotin phosphoramidite for 5'-end-labeling, affinity purification and phosphorylation of synthetic oligonucleotides. Nucleic Acids Res, 24, 361.
5. Mora N, Lacombe JM, Pavia AA. (1995) A new approach to phosphoserine, phosphothreonine and phosphotyrosine synthons and to thiophospho analogs. Stepwise synthesis of mono- and multiphosphorylated phosphopeptides related to src-protein kinase. Int J Pept Protein Res, 45, 53.
6. Boumendjel A, Miller SP. (1994) Synthesis of sphingosine-1-phosphate and dihydrosphingosine-1-phosphate. J Lipid Res, 35, 2305.
7. Kitas E, Kung E, Bannwarth W. (1994) Chemical synthesis of O-thiophosphotyrosyl peptides. Int J Pept Protein Res, 43, 146.
8. Tegge W, Ballou CE. (1992) Syntheses of D-myo-inositol 1,4,5-trisphosphate affinity ligands. Carbohydr Res, 230, 63.
9. Perich JW, Reynolds EC. (1991) Fmoc/solid-phase synthesis of Tyr(P)-containing peptides through t-butyl phosphate protection. Int J Pept Protein Res, 37, 572.
10. Lacombe JM, Andriamanampisoa F, Pavia AA. (1990) Solid-phase synthesis of peptides containing phosphoserine using phosphate tert.-butyl protecting group. Int J Pept Protein Res, 36, 275.
特别提醒:
1) 本公司所有产品仅限于专业人员用于生命科学研究,不得用于临床诊断或治疗,不得用于食品或药品,不得存放于普通住宅。
2) 本公司所有产品必须由合格专业技术人员操作同时佩戴口罩/手套/实验服并遵守生物实验室安全操作规程!
| Name | Chemical Phosphorylation Reagent I (CPR I) | ||
|---|---|---|---|
| CAT# | 6001-100mg | CAS# | N/A |
| Storage# | -20°C Sealed & desiccated & Minimized light exposure | Shelf Life# | 24 months |
| Ex(nm)# | N/A | Em(nm)# | N/A |
| MW# | 656.77 | Solvent# | MeCN,DMSO |
| Name | Chemical Phosphorylation Reagent I (CPR I) |
|---|---|
| CAT# | 6001-100mg |
| CAS# | N/A |
| Storage# | -20°C Sealed & desiccated & Minimized light exposure |
| Shelf Life# | 24 months |
| Ex(nm)# | N/A |
| Em(nm)# | N/A |
| MW# | 656.77 |
| Solvent# | MeCN,DMSO |
 详细说明书
详细说明书 技术资料
技术资料