6-FITC
英文名:6-FITC, 6-Flourescein iso-thiocyanate;FITC 'Isomer II' [6-FITC, fluorescein-6-isothiocyanate]
结构式:

Fluorescent Dye Isothiocyanates
Isothiocyanates form thioureas upon reaction with amines. It is proven that some thiourea products (in particular, the conjugates from a-amino acids/peptides/proteins) are much less stable than the conjugates that are prepared from the corresponding succinimidyl esters. It has been reported that antibody conjugates prepared from fluorescein isothiocyanates deteriorate over time. We strongly recommend that you use succinimidy esters for your conjugations whenever possible. There are few factors that need be considered when SE compounds are used for conjugation reaction:
1). Solvents: For the most part, reactive dyes are hydrophobic molecules and should be dissolved in anhydrous dimethylformamide (DMF) or dimethylsulfoxide (DMSO).
2). Reaction pH: The labeling reactions of amines with isothiocyanates are strongly pH dependent. Isothiocynate reagents react with non-protonated aliphatic amine groups, including the terminal amines of proteins and the e-amino groups of lysines. Protein modifications by isothiocyanates may require a pH 9.0-10.0 for optimal conjugations.
3). Reaction Buffers:Buffers that contain free amines such as Tris and glycine must be avoided when using an amine-reactive reagent. Ammonium salts (such as ammonium sulfate and ammonium acetate) that are widely used for protein precipitation must also be removed before performing dye conjugations. High concentrations of nucleophilic thiol compounds should also be avoided because they may react with the labeling reagent to form unstable intermediates that could destroy the reactive dye.
4). Reaction Temperature: Isothiocynate conjugations are usually done at room temperature. However, either elevated or reduced temperature may be required for a particular labeling reaction.
FITC 'Isomer II' [6-FITC, fluorescein-6-isothiocyanate]
Cat# | Size | Price | MW | Abs | Em | Soluble in | Storage |
125 | 100 mg | $49 | 389.38 | 494 nm (pH>9.0) | 520 nm (pH>9.0) | DMSO or DMF | 4 oC and desiccated |
Features and Biological Applications
5-FITC and 6-FITC have very similar absorption and fluorescence spectra. However, the isomers may differ in their binding and reactivities to proteins, and the conjugates may migrate differently in electrophoretic gels. Thus, we offer highly purified single isomers. Cautions must be exercised for the storage of FITC conjugates.
References
1. Balogh, P., et al., Hapten-mediated identification of cell membrane antigens using an anti-FITC monoclonal antibody. J Immunol Methods 1994, 169, 35-40.
2. Korting, H.J. and E. Schubert, Preparation of FITC-labeled permanent preparations for fluorescent antibody technic]. Z Med Lab Diagn 1977, 18, 387.
3. Gunduz, N., The use of FITC-conjugated monoclonal antibodies for determination of s-phase cells with fluorescence microscopy. Cytometry 1985, 6, 597-601.
| Name | 6-FITC;6-Flourescein iso-thiocyanate | ||
|---|---|---|---|
| CAT# | 122-100mg;122-1g | CAS# | 890090-49-0 / 3326-31-6 |
| Storage# | −20°C干燥避光 | Shelf Life# | 12个月 |
| Ex(nm)# | 494 | Em(nm)# | 520 |
| MW# | 389.38 | Solvent# | DMSO |
| Name | 6-FITC;6-Flourescein iso-thiocyanate |
|---|---|
| CAT# | 122-100mg;122-1g |
| CAS# | 890090-49-0 / 3326-31-6 |
| Storage# | −20°C干燥避光 |
| Shelf Life# | 12个月 |
| Ex(nm)# | 494 |
| Em(nm)# | 520 |
| MW# | 389.38 |
| Solvent# | DMSO |

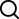
 Specification
Specification Support
Support



